Sirolimus coated balloon
Bio-sphere technology
The Latest Generation of Drug Coated Balloon
Bio-sphere technology
BIO-SPHERE TECHNOLOGY
Sirolimus:
- 1.27 µg/mm²
- Cytostatic effect
- Drug most widely used for Drug Eluting Stents
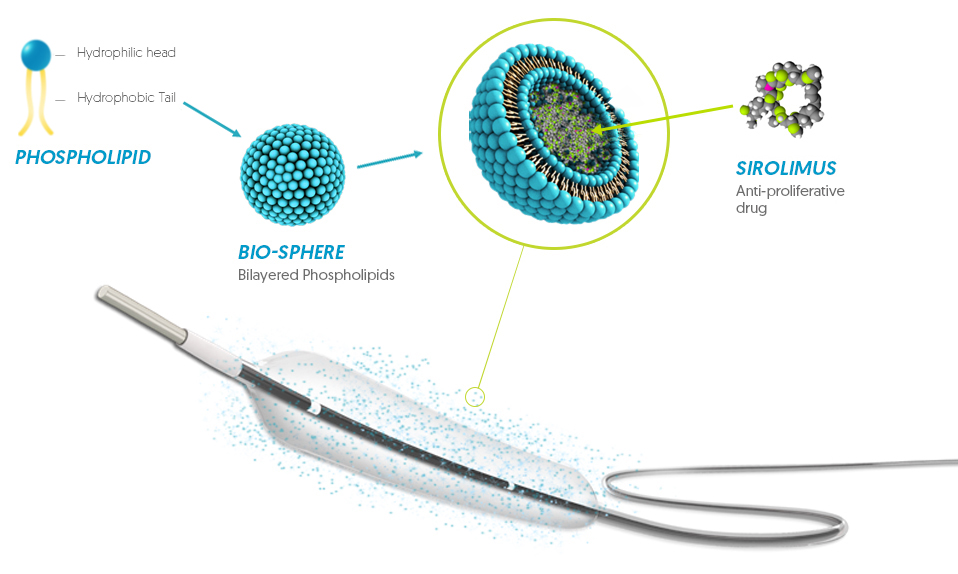
Cell-Membrane Bio-Mimicking Carrier
Small Size for Higher Uptake
Bio-Sphere Drug Encapsulation
Two-steps release
- Better compatibility & acceptance in tissue
- Promotes healing and assists in endothelialization
Small Size for Higher Uptake
- Easier & faster transfer into arterial tissue
Bio-Sphere Drug Encapsulation
- Stabilization & protection
Two-steps release
- Bio-spheres transfer into tissue
- Drug release from bio-spheres
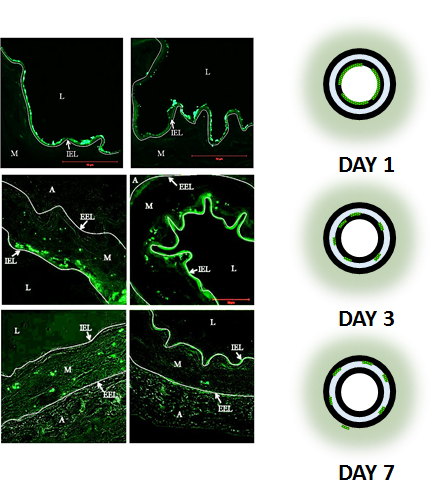
Homogeneous Drug Repartition within 7 days
COATING PROCESS

Effective Adhesion to Balloon Surface
Uniform Surface Coating
- No coating delamination
- Low "particulate" generation
Uniform Surface Coating
- Homogeneous drug distribution
- Protection during transit (between the folds)



* data on file
RELIABLE DRUG DELIVERY PLATFORM
- 5F Guiding Catheter Compatible
- Distal Shaft Hydrophilic Coating
- 0,016” Entry Profile
- Semi-Compliant Balloon material

Yangtze µ catheter system physicians feedback: 642 cases*
* data on file
PRE-CLINICAL DATA
Safety and Efficacy verified
Histological Evaluation after 28 days implantation*
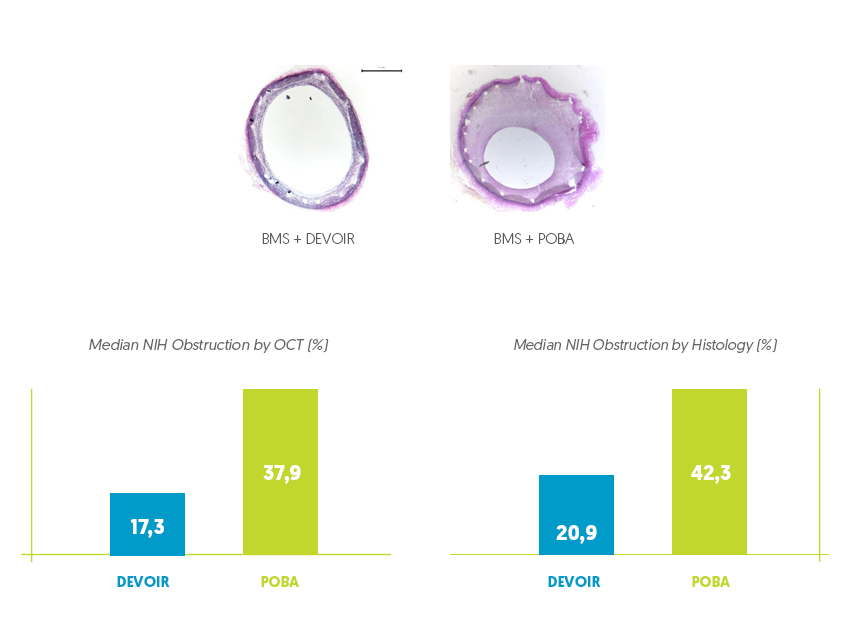
Effective reduction of neo-intimal hyperplasia

Effective reduction of neo-intimal hyperplasia
* data on file
Pharmacokinetic evaluation*
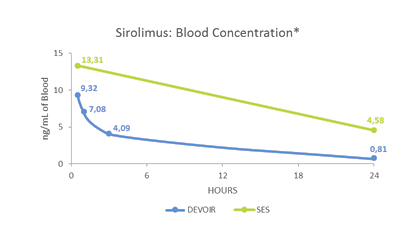
- Low Sirolimus blood levels
- Lower blood levels compared to Sirolimus Eluting Stent (SES)
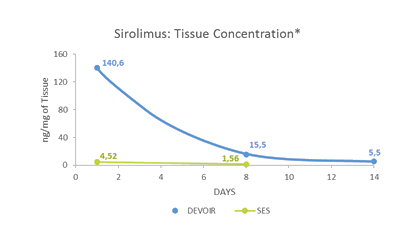
- Sirolimus retention up to 14 days
- Higher concentration compared to Sirolimus Eluting Stent (SES)
* data on file
Clinical Data
Observational study for safety and performance demonstration
12 months cumulative clinical results
Overall population (n=347)
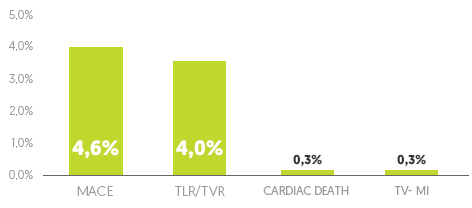
Overall population (n=347)

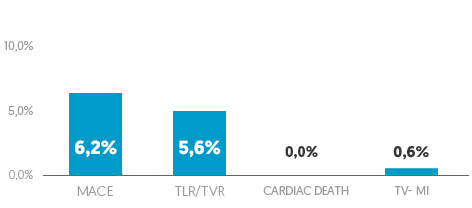
ISR subgroup analysis (n=162)

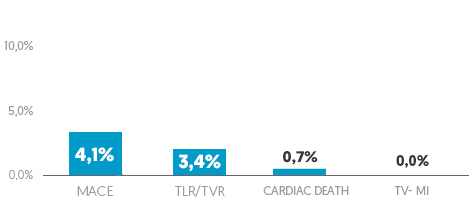
Small vessel subgroup analysis (n=148)

Specifications
Indications
The DEVOIR Sirolimus coated balloon is indicated to dilate the diseased segment(s) and deliver drug in a coronary artery to improve myocardial perfusion with lesion lengths ranging from 8 to 38 mm and diameters ranging from 1.50 mm to 4.00 mm.
The DEVOIR Sirolimus coated balloon is indicated to dilate the diseased segment(s) and deliver drug in a coronary artery to improve myocardial perfusion with lesion lengths ranging from 8 to 38 mm and diameters ranging from 1.50 mm to 4.00 mm.
| Drug | Sirolimus |
|---|---|
| Drug dose | 1.27 µg/mm² |
| Drug carrier | Phospholipid based excipient |
| Balloon material | Polyamide blend (Semi-compliant balloon) |
| Catheter design | Rapid Exchange design |
| Shaft diameter - Proximal | 1.7 F |
| Shaft diameter – Distal | 2.5 F |
| Usable catheter length | 140 cm |
| Tip profile | 0.016" |
| NP* | 6 bar |
| RBP* | 16 bar (14 bar for 4.00 / 25 to 40 mm) |
| Guiding catheter compatibility | 5F (0.058" ID) for all sizes |
| Guidewire compatibility | 0.014" maximum recommended |

Ordering information
| LENGTH (mm) | DIAMETER (mm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1.50 | 2.00 | 2.25 | 2.50 | 2.75 | 3.00 | 3.25 | 3.50 | 4.00 | ||||||
| 10 | CDE15010 | CDE20010 | CDE22510 | CDE25010 | CDE27510 | CDE30010 | CDE32510 | CDE35010 | CDE40010 | |||||
| 15 | CDE15015 | CDE20015 | CDE22515 | CDE25015 | CDE27515 | CDE30015 | CDE32515 | CDE35015 | CDE40015 | |||||
| 20 | CDE15020 | CDE20020 | CDE22520 | CDE25020 | CDE27520 | CDE30020 | CDE32520 | CDE35020 | CDE40020 | |||||
| 25 | CDE15025 | CDE20025 | CDE22525 | CDE25025 | CDE27525 | CDE30025 | CDE32525 | CDE35025 | CDE40025 | |||||
| 30 | CDE15030 | CDE20030 | CDE22530 | CDE25030 | CDE27530 | CDE30030 | CDE32530 | CDE35030 | CDE40030 | |||||
| 35 | CDE15035 | CDE20035 | CDE22535 | CDE25035 | CDE27535 | CDE30035 | CDE32535 | CDE35035 | CDE40035 | |||||
| 40 | CDE15040 | CDE20040 | CDE22540 | CDE25040 | CDE27540 | CDE30040 | CDE32540 | CDE35040 | CDE40040 | |||||
 Download Brochure
Download Brochure

